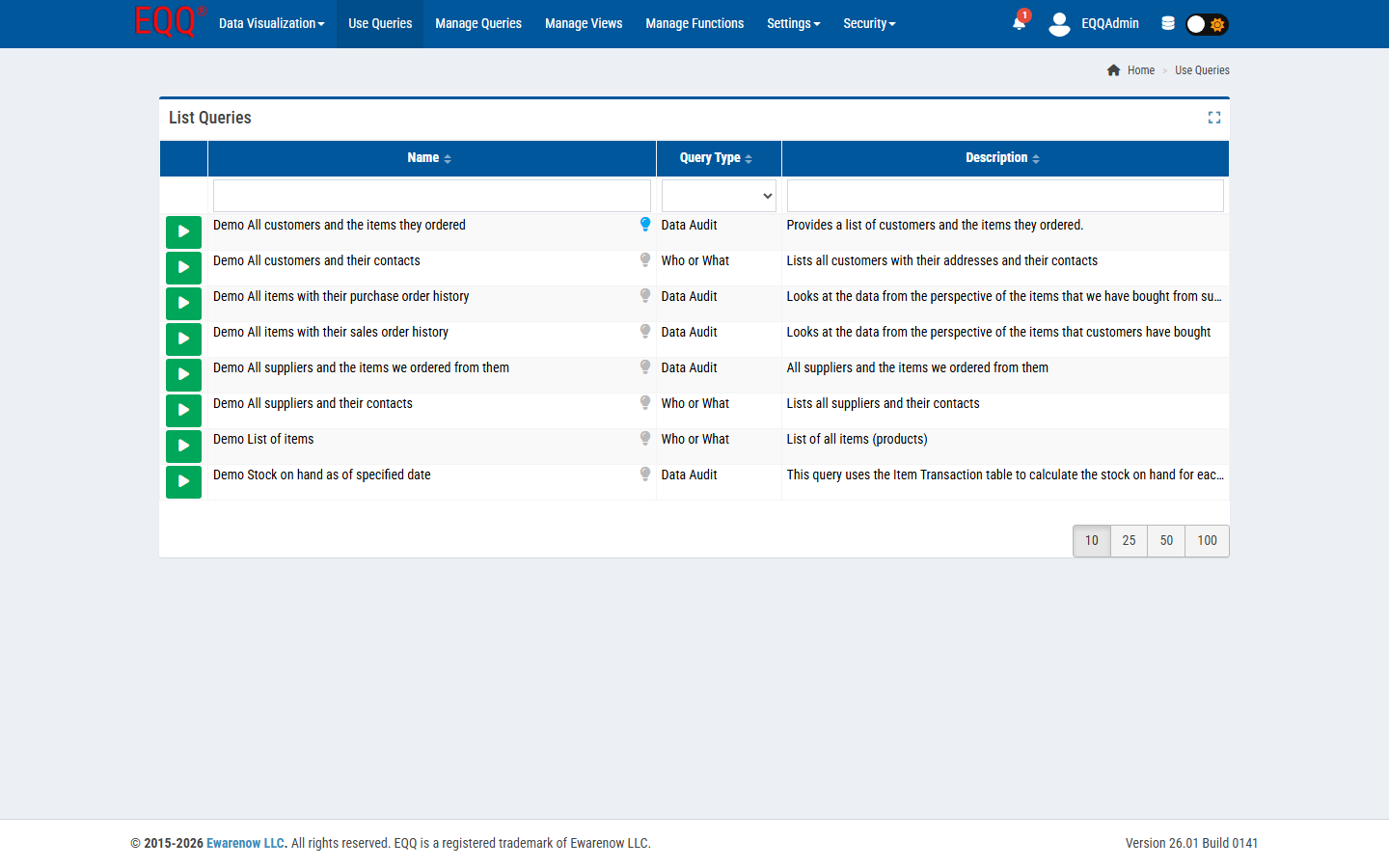
Labs have a very specific shape of recurring question: “give me every sample above the MDL for parameter X in the last reporting period”. Here are five of the most common, pre-built for EQQ.
1. Samples exceeding MDL by analytical method: flag every result above the detection threshold
This query returns every sample result where the measured concentration exceeds the Method Detection Limit for the associated analytical method. It groups results by method and analyte so supervisors can immediately see which parameters are generating exceedances and in what volume. Running this query before a reporting deadline lets QA staff review borderline results before they reach the client. Because the query is stored as an Active EQQ query, the same logic is applied consistently every run, eliminating analyst-to-analyst variation in the cutoff calculation.
2. QC out-of-control: identify quality control samples outside acceptable accuracy and precision limits
Laboratory quality control relies on method blanks, matrix spikes, and duplicates all falling within defined acceptance windows. This query surfaces every QC sample from the current reporting period whose result falls outside those windows — high or low. Finding out-of-control QC early gives analysts time to investigate the cause, re-run affected samples if needed, and document corrective action before the batch is reported. Storing this query in EQQ with an Active lifecycle state means the audit trail records every time a QC review was performed and by whom.
3. Turnaround time by analyst or method: measure how long samples take from receipt to final result
Turnaround time is a contractual commitment in most laboratory service agreements. This query calculates elapsed time between sample receipt and the date of final result entry, grouped by analyst or by analytical method. Outliers — batches or analysts consistently running long — surface immediately so lab managers can reallocate work or investigate bottlenecks. Because the query accepts a date-range parameter, it can be re-run weekly for trending without any SQL knowledge on the part of the lab manager.
4. Chain-of-custody breaks: detect samples with missing custody signatures or timestamps
Regulatory bodies require an unbroken documented chain of custody from sample collection through disposal. This query identifies samples where a custody transfer record is missing a signature, a timestamp, or a required custodian entry. Even a single gap can invalidate a data package during an audit. Running this query at sample receipt — and again before reporting — allows the lab to contact field collectors for corrections while the information is still accessible, rather than discovering the gap during a third-party audit.
5. Client pull-list for the current period: all samples due for client reporting right now
At period end, labs need a complete list of every sample associated with a client work order that is ready — or overdue — for reporting. This query returns all samples whose analytical work is complete and whose results have not yet been delivered to the client, scoped to the current reporting period. It acts as the master checklist that drives the data package assembly process. Locking this query in EQQ ensures the definition of "ready for reporting" is consistent across every reporting cycle and that every pull is logged for audit purposes.
Each query uses EQQ Functions like fQQ_ReportingPeriod(@AsOf) and fQQ_MDLCompare(@result, @mdl) so the math is consistent across the lab.
Compliance bonus
All five queries are marked Active and audited. If an external auditor asks how you computed “percent exceeding MDL”, the answer is: “we ran query #12, here is the SQL, here is the run log, here is the exported file.”
Key Terms
- MDL - Method Detection Limit - the lowest concentration reliably detected by the analytical method.
- QC - Quality Control - the process of verifying that test results meet predefined accuracy and precision criteria.
- TAT - Turn-Around Time - the elapsed time between sample receipt and final result reporting.
- COC - Chain of Custody - the documented sequence of possession, transfer, and handling of a sample from collection to disposal.